Yoguely is reader-supported. When you buy through links on our site, we may earn an affiliate commission. Learn more
How to Use Soap and Detergent The Right Way

In today’s post I’m going to show you the exactly how to use surfactants, like soap and detergent, the right way to clean and even disinfect surfaces from certain microbes like COVID-19.
In fact, this is the same process I use to safely clean with soap and detergent.
So if you would like to know “How to clean with detergent or soap and water?”, you’ll love this guide.
Let’s jump into it!
Why Are Soaps and Detergents Good for Cleaning?
Both detergent and soap are common cleaning agents that are made with surfactants. In detail, surfactants are compounds that lower the surface tension between a liquid and another material in a solid, liquid, or gas state.[2]
When used for cleaning, surfactants helps solubilize grime. In other words, it dissolves the dirt ingrained on surfaces.[3] Which then makes it really easy to wash off.
Cool huh?
Well, surfactants can do more than just dissolve grime, they can also inactivate certain viruses and bacteria. More on that soon.
What is Soap and Detergent Good For?
Material it helps remove: Both soap and detergent help remove dirt and oils so that you can easily wash it away with running water.[4]
Surfaces you can clean: Use soap on your skin. Conversely, use appropriate detergents for surfaces where you do not want soap scum to form. For instance, your hair, laundry, floors, counter tops, and other surfaces. I’ll explain more about soap scum soon.
Is Soap and Detergent a Disinfectant?
The Environmental Protection Agency (EPA) does not register soaps and detergents as a disinfectant. Although soap and detergent can inactivate some bacteria, they do not meet the requirement of killing 99.99% of germs to be considered a disinfectant.[32]
If you wish to use an EPA approved disinfectant, check out sodium hypochlorite (bleach), isopropyl alcohol, or hydrogen peroxide.

How Do Soap and Detergent Kill Viruses and Bacteria?
Surfactants, like those in soap and detergent, kills microorganisms by disorganizing their membrane lipid bilayer.[4] To explain, the lipid bilayer is what makes up the cell membrane of almost all life forms.
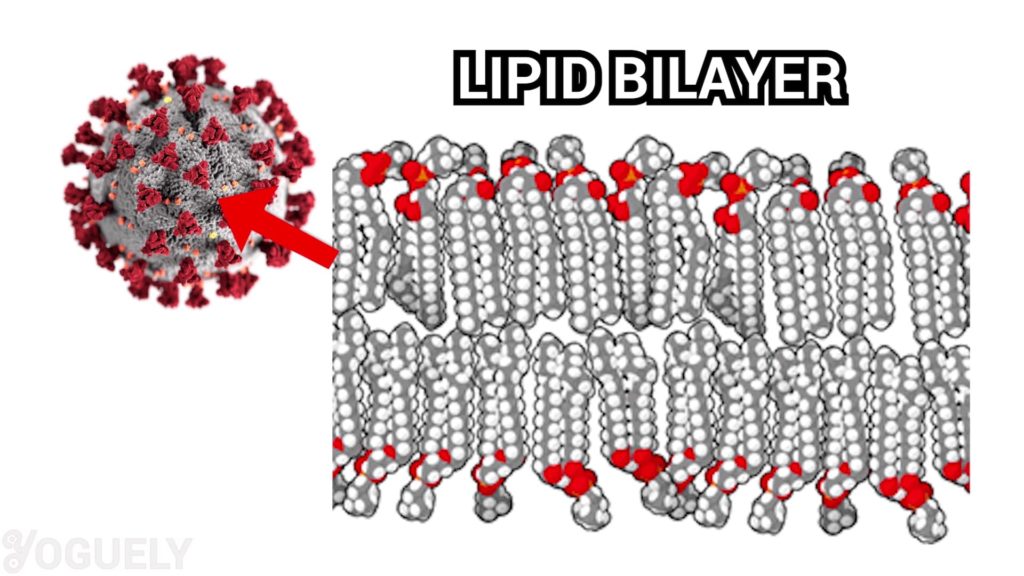
Therefore, surfactants break down the cell membrane of organisms and many viruses.[5]
Not only that, surfactants also unfold the proteins of microorganisms, solubilizing it, and destructuring it so it is no longer in the right shape to function.[6]
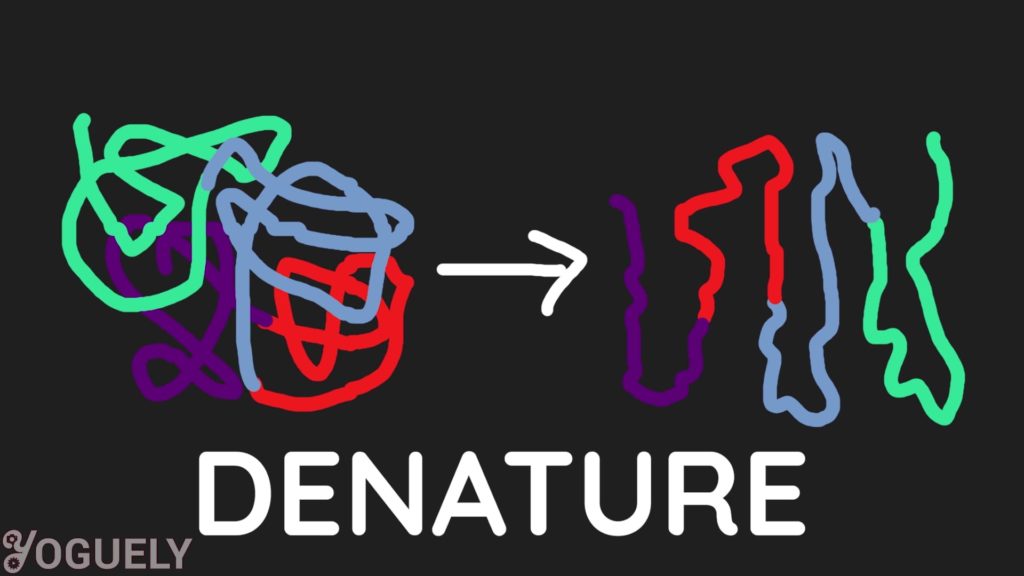
This process is called denaturation. By denaturing the proteins in a living cell, it either disrupts cell activity or kills the cell completely.[7]
Can Soap and Detergent Kill Coronavirus?
Yes, by the same token soap and detergent can breakdown the coronavirus species, including the novel COVID-19 virus. Specifically because coronaviruses have a lipid envelope.
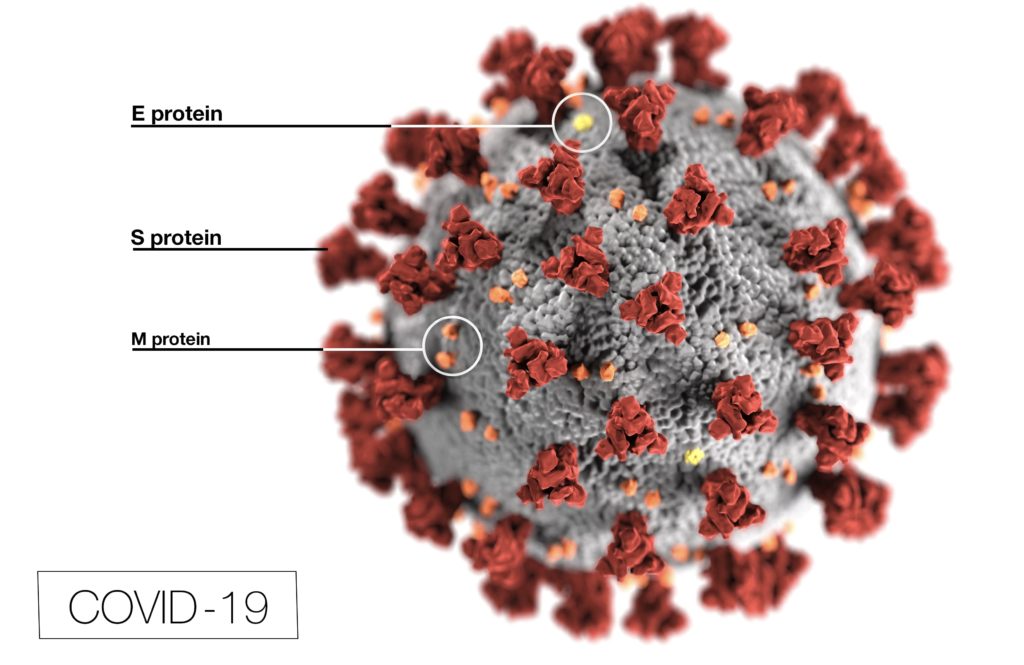
Surfactants disactivates the coronavirus by dissolving the layer of fat, the lipid bilayer, that makes up the cell membrane. Without a casing, the virus’s genetic material remains disfunctional.[8],[9],[10],[11]
Antibacterial Soap or Plain Soap: Which Is Most Effective Against Coronavirus?
Antibacterial soap has added chemicals that help inhibit the growth or kill of many kinds of bacteria. Albeit, with respect to cleaning, meaning to reduce the quantity of bacteria and viruses on a surface, antibacterial soap is no better than plain soap.[18],[4],[19],[20]
Furthermore, the downside of antibacterial soap is that it encourages bacteria to develop antibiotic resistance. These newly resistant microbes, also known as superbugs, require very expensive and highly toxic amounts of antibiotics.[21]
Lastly, antibacterial soap may have detrimental long-term effects on your health.

So long as there are adverse health effects to using antibacterial soap and no added health benefit according to the CDC,[22] the choice is simple, save your cash. Stick to using plain soap and water.
What is the Difference Between Soap and Detergent?
Soap is made by mixing fats and oils with a base. While detergent is made by mixing chemical compounds.[4]

Given that soap is made of fatty acids, soap tends to react with minerals in hard water, forming soap scum. This soap scum is insoluble, so it will not dissolve in water. And it is really hard to remove once it build up and hardens.[13],[14]
If you have soap scum on surfaces, just apply the classic low-cost cleaning ingredients, baking soda and vinegar, to help separate it and wash it off.[15]
This is part of the reason why we do not use soap to clean surfaces like floors and countertops.
Furthermore, scum is the big reason why we do not use soap on our scalp and hair. It can cause itchiness and lead to seborrheic dermatitis.[32] Instead we use shampoo (paid link) which is a detergent specially made to use on your hair.

Unlike soaps, detergent does not form scum because detergent is made from propene, which comes from distilling unrefined petroleum. When detergent reacts with minerals in hard water, the new substance formed is soluble, thus dissolves in water.[14]
Which Is Better, Soap or Detergent?
Soap and detergents both have their purpose. Now I’ll compare which one is better when it comes to biodegradability, skin care, and for cleaning.
Biodegradability
With respect to biodegradability, soap molecules easily break down to simple atoms: carbon, hydrogen, and oxygen. Which, can then be readily consumed by plants and microorganisms.
Provided that detergents are a petroleum-derived product, they take much longer to breakdown. As a result, it can be very harmful for the ecosystem. Because of detergents, you can oftentimes see foaming in sewage treatment plants and waterways.
The complex molecules also lead to eutrophication of waterways. Having excess nutrients in water makes plant life grow rapidly. In effect, depleting the oxygen supply in the water, and overcoming animal life nearby.[16]
Skin Care
When it comes to skin care, detergents tend to irritate skin and cause dryness. It can even cause skin reactions for some people.
On the other hand, soap tends to be better for most people’s skin.[14]
One of my favorite hand and body soap is African black soap (paid link). It is great on oily skin and even has natural antimicrobial properties.[17]

Surfaces, Dishwashing, and Laundry Cleaner
As opposed to soap, detergents are more soluble in hard water and don’t create soap scum. Consequently, making them very effective for removing dirt from surfaces, dishes, laundry, and even hair.
Soap and Detergent Safety Measures
Here are some safety measure to take whenever using soap and detergent.
Detergent
MSDS: There are many kinds of detergent solutions made for all sorts of applications. So you’ll need to read the safety data sheet for the one you are using.
For example, this is the MSDS sheet for Persil Proclean Power-Liquid laundry detergent (paid link).
Hazards: In general, detergents are harmful to your skin and eyes, causing severe irritation if you get contaminated with it.
Personal Protective Equipment (PPE): With this in mind, whenever handling detergents, you’ll want to wear personal protective equipment (PPE) to keep safe in an accident. Simply wear splash-proof safety goggles (paid link), protective clothing for your skin, and chemical-resistant gloves (paid link).

Soap
MSDS: Again, there are many soaps made out there so find the MSDS for the soap you are using.
For example, this is the MSDS for a African black soap.
Hazards: Soap may irritate your eyes. And if you apply it for too long on your skin, you may develop redness, skin sensitization, or dermatitis.[30]
I developed dermatitis on my fingers from washing dishes without wearing appropriate PPE — gloves.
Big mistake.
Now whenever I wash my hands with soap too much my fingertips gets really dry and, in the winter, they tend to crack open.
Personal Protective Equipment (PPE): Avoid using soap near your eyes or use safety googles if you need to. Use soap appropriately so that it doesn’t stay on your skin for too long. More on that next.
How to Wash with Soap or Detergent
To rid surfaces of dirt and microorganisms, clean with soap or detergent. This is an important step before disinfecting.
When using detergents, follow label directions for safe and effective use.
To clean with soap, follow these five steps:[23]
- Wet with running water at warm or cold temperatures.[19]
- Apply soap by lathering it with liberal amounts of frothy bubbles.
- Scrub for at least 20 seconds.
- Rinse with running water.
- Dry using a towel or by air drying.
Do not use hot water to wash your hands. Studies show that if there are surfactants on your skin, the amount of skin damage you can get increases as water temperature increases.[31]
You can also apply this knowledge when taking showers too. Do yourself a favor. Take quick showers with warm or cold water. You’ll save on water, heater costs, and also prevent developing irritant contact dermatitis.
How Long Should Soap or Detergent Sit on Surface to Disinfect?
As you see in the 5 steps to cleaning with soap, there isn’t a set contact time for which the surfaces must remain wet with soap and water.
The real key step is scrubbing. The mechanical action of scrubbing is what will help “unstick” most of the dirt on surfaces. So that you can then rinse it away with water.
After you’ve cleaned to remove as much dirt and microorganisms as you can, the next step is to use a disinfectant to kill the remaining bacteria and viruses on the surface.
Disinfectants do need to meet a required contact time to be effective.
Should You Use Soap or Detergent to Clean Fruits and Vegetables?
Vegetables and fruits are porous so they have little tiny voids where matter can get trapped inside.[26] Which means those holes could be filled with gasses, contaminated droplets, or dirt.
Soap and detergents do make it easier to remove debris. However, both the CDC and the FDA recommends washing or scrubbing fruits and vegetables under running water.[24],[25],[26]
They do not make any recommendations to use soap or detergent, and for a good reason. Even after you rinse it off from produce, it is possible for some of the cleaning agents to remain trapped in the pores.
You definitely want to avoid eating detergent since it is quite poisonous if ingested.
With regards to soap, it is generally less toxic than detergent. However, ingesting soap in large quantities could cause gastrointestinal irritation, vomiting, and diarrhea.[28]
If you do not eat the skin, having trace amounts of soap trapped in pores might be a minor problem. That is, compared to for instance, having active coronavirus on the skin. So assess your risks accordingly.
What is the Shelf Life of Soap and Detergent?
Shelf life: Typically, Soap and detergent do not have a printed expiration date “EXP” or a “best before” date.
On the contrary, those with enzyme[29] additives or those with anti-microbial ingredients, such as triclosan and triclocarban, may have an expiration date since they lose their effectiveness over time.
How to Store Soap for Long Term?
Elements like ultraviolet rays and heat can break down chemical bonds.[29] You can protect it by carefully storing soap away from sunlight, and temperature extremes. If done so, simple soap and detergent could still be effective for well over 3 years.
Conclusion
There you have it folks. That’s my guide on how to use surfactants, like soap and detergent, the right way.
Leave your thoughts in the comment section below. Or join the discussion in the Yoguely Community Forum.
I’m Aida Yoguely. Thanks for learning with me today. And I’ll see you soon.
In my next post, learn about vinegar and how to use it for cleaning and disinfecting. To stay tuned, join our newsletter and get the latest content straight to your inbox.
Video
Be sure to subscribe and hit the notification bell to stay tuned for the latest videos.
References
2. ^ (May 22, 2020) “Surfactant” Wikipedia. Retrieved May 23, 2020.
3. ^ James Thomson (2012) “Solubilization” Handbook of Radioactivity Analysis. Retrieved May 23, 2020.
32. ^ “List N: Disinfectants for Use Against SARS-CoV-2”. EPA. Retrieved May 27, 2020.
4. ^ (May 14, 2020). “Soap”. Wikipedia. Retrieved May 23, 2020.
5. ^ (May 21, 2020). “Liquid Bilayer”. Wikipedia. Retrieved May 23, 2020.
6. ^ (May 17, 2020). “Protein Folding”. Wikipedia. Retrieved May 23, 2020.
7. ^ (May 8, 2020). “Denaturation (biochemistry)”. Wikipedia. Retrieved May 23, 2020.
8. ^ Cascella M, Rajnik M, Cuomo A, et al. (Apr 6, 2020) “Features, Evaluation and Treatment Coronavirus (COVID-19)” In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-.
9. ^ “Guidance at a Glance – IPC for Coronavirus”. Linclnshire County Council. Retrieved May 23, 2020.
10. ^ (May 21, 2020). “COVID-10: infection prevention and control (IPC)”. Public Health England GOV.UK. Retrieved May 23, 2020.
11. ^ (September 2016). “Middle East Respiratory Syndrome (MERS-CoV) Infection Prevention and Control”. Public Health England. Retrieved May 23, 2020.
13. ^ Draelos Z. D. (2010). Essentials of Hair Care often Neglected: Hair Cleansing. International journal of trichology, 2(1), 24–29. https://doi.org/10.4103/0974-7753.66909
14. ^ “About Detergents”. Royal Society of Chemistry. Retrieved May 23, 2020.
15. ^ (April 22, 2020). “How to Clean Soap Scum off Every Bathroom Surface”. the spruce. Retrieved May 23, 2020.
16. ^ Bajpai D, Tyagi VK. Laundry detergents: an overview. J Oleo Sci. 2007;56(7):327‐340. doi:10.5650/jos.56.327
17. ^ (March 12, 2020). “African Black Soap”. Wikipedia. Retrieved May 26, 2020.
18. ^ (April 27, 2020). “Antibacterial soap”. Wikipedia. Retrieved May 26, 2020.
19. ^ “Handwashing at Home, at Play, and Out and About”. CDC. Retrieved May 26, 2020.
20. ^ Aiello, A. E., Larson, E. L., & Levy, S. B. (2007). Consumer antibacterial soaps: effective or just risky?. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America, 45 Suppl 2, S137–S147. https://doi.org/10.1086/519255
21. ^ (May 21, 2020). “Antimicrobial resistance”. Wikipedia. Retrieved May 26, 2020.
22. ^ (April 23, 2020). “Handwashing: Clean Hands Save Lives: Publications, Data, & Statistics”. CDC. Retrieved May 26, 2020.
23. ^ (April 2, 2020). “When and How to Wash Your Hands”. CDC. Retrieved May 26, 2020.
24. ^ (February 12, 2020). “Fruit and Vegetable Safety”. CDC. Retrieved May 26, 2020.
25. ^ (June 10, 2018). “7 Tips for Cleaning Fruits, Vegetables”. FDA. Retrieved May 26, 2020.
26. ^ Nugraha, B., Verboven, P., Janssen, S., Wang, Z., & Nicolaï, B. M. (2019). Non-destructive porosity mapping of fruit and vegetables using X-ray CT. Postharvest biology and technology, 150, 80-88.
27. ^ “Steps to Safe and Healthy Fruits and Vegetables”. CDC. Retrieved May 26, 2020.
28. ^ CANN HM, VERHULST HL. Toxicity of household soap and detergent products and treatment of their ingestion. Am J Dis Child. 1960;100:287‐290. doi:10.1001/archpedi.1960.04020040289022
29. ^ Estell, D. A. (1991). U.S. Patent No. 5,039,446. Washington, DC: U.S. Patent and Trademark Office.
29. ^ Why does ultraviolet light cause color to fade?. Library of Congress. Retrieved June 3, 2020.
30. ^ Larson, E. (2001). Hygiene of the Skin: When Is Clean Too Clean?. Emerging Infectious Diseases, 7(2), 225-230. https://dx.doi.org/10.3201/eid0702.700225.
31. ^ Larson, E. (Berardesca E, Vignoli GP, Distante F, Brizzi P, Rabbiosi G. Effects of water temperature on surfactant-induced skin irritation. Contact Dermatitis. 1995;32(2):83‐87. doi:10.1111/j.1600-0536.1995.tb00751.x
32. ^ D’Souza P, Rathi SK. Shampoo and Conditioners: What a Dermatologist Should Know?. Indian J Dermatol. 2015;60(3):248‐254. doi:10.4103/0019-5154.156355
- Why Helicopter Money Is Inflationary - 2021-10-09
- How Interest Rates Affect Inflation - 2021-10-08
- How Long Has Inflation Existed? - 2021-10-01




